Professor Lei Chang's and Professor Zengli Zhang's research groups jointly published a research paper titled “Autophagy induced by mechanical stress sensitizes cancer cells to ferroptosis by NCOA4-FTH1 axis” in the journal Autophagy (DOI: 10.1080/15548627.2025.2469129). This study reveals the molecular mechanism through which mechanical stress regulates ferroptosis sensitivity via NCOA4-mediated ferritinophagy of FTH1, highlighting the central role of the mechanical stress in the regulation of ferroptosis and providing new research perspectives for clinical radiotherapy.
Previous studies have shown that cell density can affect ferroptosis sensitivity. For example, cadherin, which is dependent on cell density, can regulate the expression of genes such as ACSL4 and TFRC through the NF2-HIPPO pathway, thereby influencing cellular susceptibility to ferroptosis. Additionally, in a high cell density environment, neutral triacylglycerols (TAG) rich in polyunsaturated fatty acids (PUFA) can accumulate extensively, making cells more prone to ferroptosis. However, these studies primarily focused on the impact of cell density on ferroptosis, whereas this research further explores the role of the mechanical stress in cellular ferroptosis sensitivity. The study found that alterations in the mechanical conditions significantly affect FTH1 levels, thereby regulating intracellular free iron content and ultimately influencing the ferroptosis sensitivity of tumor cells, which unveils the complex interplay between the mechanical stress and ferroptosis.
Intracellular free iron ions are primarily regulated by transferrin receptor (TFR), ferroportin 1 (FPN1), and ferritin. Ferritin, composed of ferritin light chain (FTL) and ferritin heavy chain (FTH), can store up to 4,000 iron atoms and maintains intracellular iron homeostasis mainly through the autophagy pathway. In this process, NCOA4 acts as a selective cargo receptor for ferritin, mediating ferritinophagy through phase separation, which is a key mechanism for maintaining intracellular iron homeostasis. This study found that under low mechanical stress conditions, NCOA4 levels significantly decrease, and the co-localization of NCOA4 and FTH1 during phase separation is reduced, ultimately affecting ferritinophagy and its downstream iron metabolism pathways. This discovery further reveals the complex relationship between iron metabolism, autophagy, FTH1 phase separation, and ferroptosis, providing a new theoretical basis for the regulation of ferroptosis by mechanical signaling pathways. In nude mouse xenograft tumor models, the research team further investigated the synergistic effects of the mechanical stress and ferroptosis induction. The results showed that combining tumor stiffening agents (which enhance the mechanical strength of tumor tissue) with ferroptosis inducers or radiotherapy significantly inhibited tumor growth and reduced tumor weight. During the rapid proliferation phase of tumor tissue, the interior of solid tumors often exhibits a high mechanical stress microenvironment due to extracellular matrix remodeling and collagen fiber deposition. This phenomenon suggests that combining ferroptosis inducers with conventional radiotherapy may be an effective strategy for improving the treatment efficacy of solid tumors.
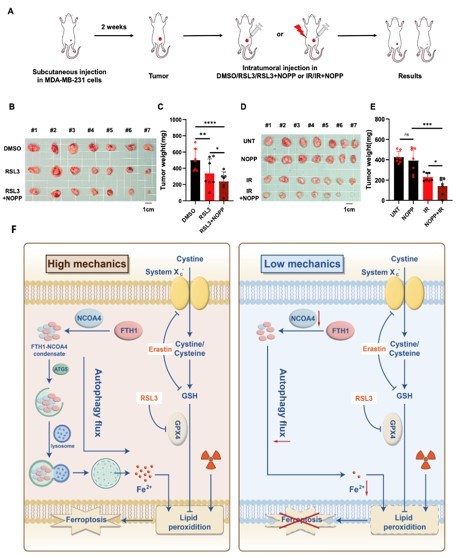
In summary, this study systematically reveals the critical role of the mechanical stress in regulating ferroptosis and indicates that NCOA4-mediated FTH1 ferritinophagy may be a core mechanism regulating ferroptosis sensitivity, offering new intervention insights and potential therapeutic strategies for radiotherapy.
The first authors of this study include Chenyu Luo (Class of 2020 master's student), Haisheng Liang(Class of 2022 master's student), postdoctoral researcher Mintao Ji, and Associate Researcher Caiyong Ye from the School of Radiation Medicine and Protection at Soochow University. Professor Lei Chang and Professor Zengli Zhang are the corresponding authors of this study. This work was supported by the National Natural Science Foundation of China, the Jiangsu Provincial Outstanding Youth Foundation, the State Key Laboratory of Radiation Medicine and Radiation Protection at Soochow University, the Jiangsu Provincial Excellent Postdoctoral Program, and the Jiangsu Provincial Natural Science Foundation.

